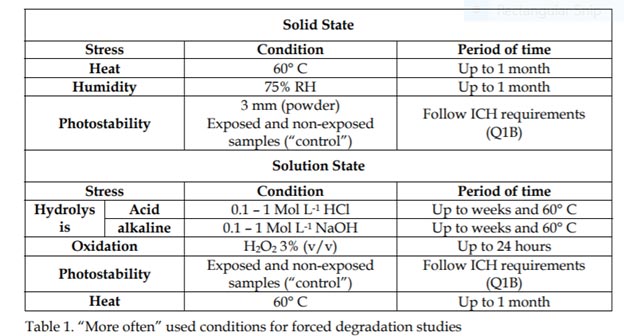
Scientific and Regulatory Policy Committee Points to Consider: Biological Sample Retention From Nonclinical Toxicity Studies - Carole E. Harbison, Adam D. Aulbach, Bindu M. Bennet, Molly H. Boyle, Mary E. Carsillo, Torrie
Compliance Policy for the Quantity of Bioavailability and Bioequivalence Samples Retained Under 21 CFR 320.38(c)