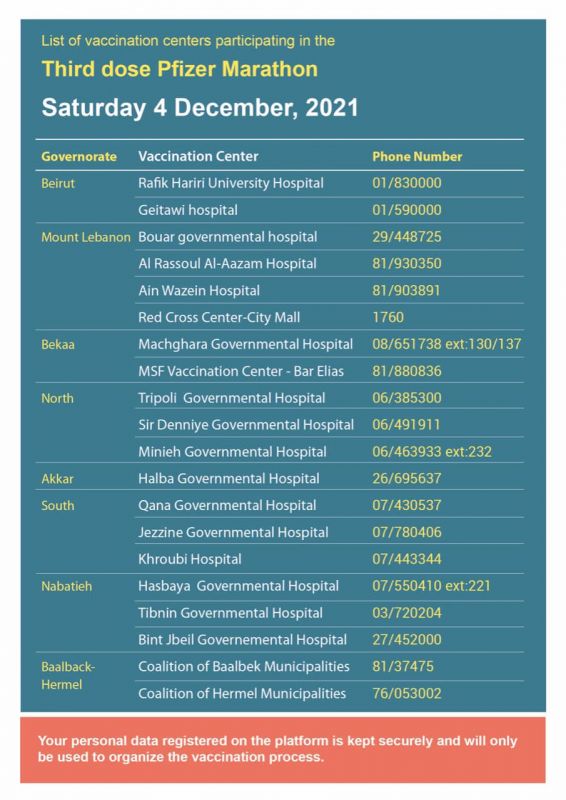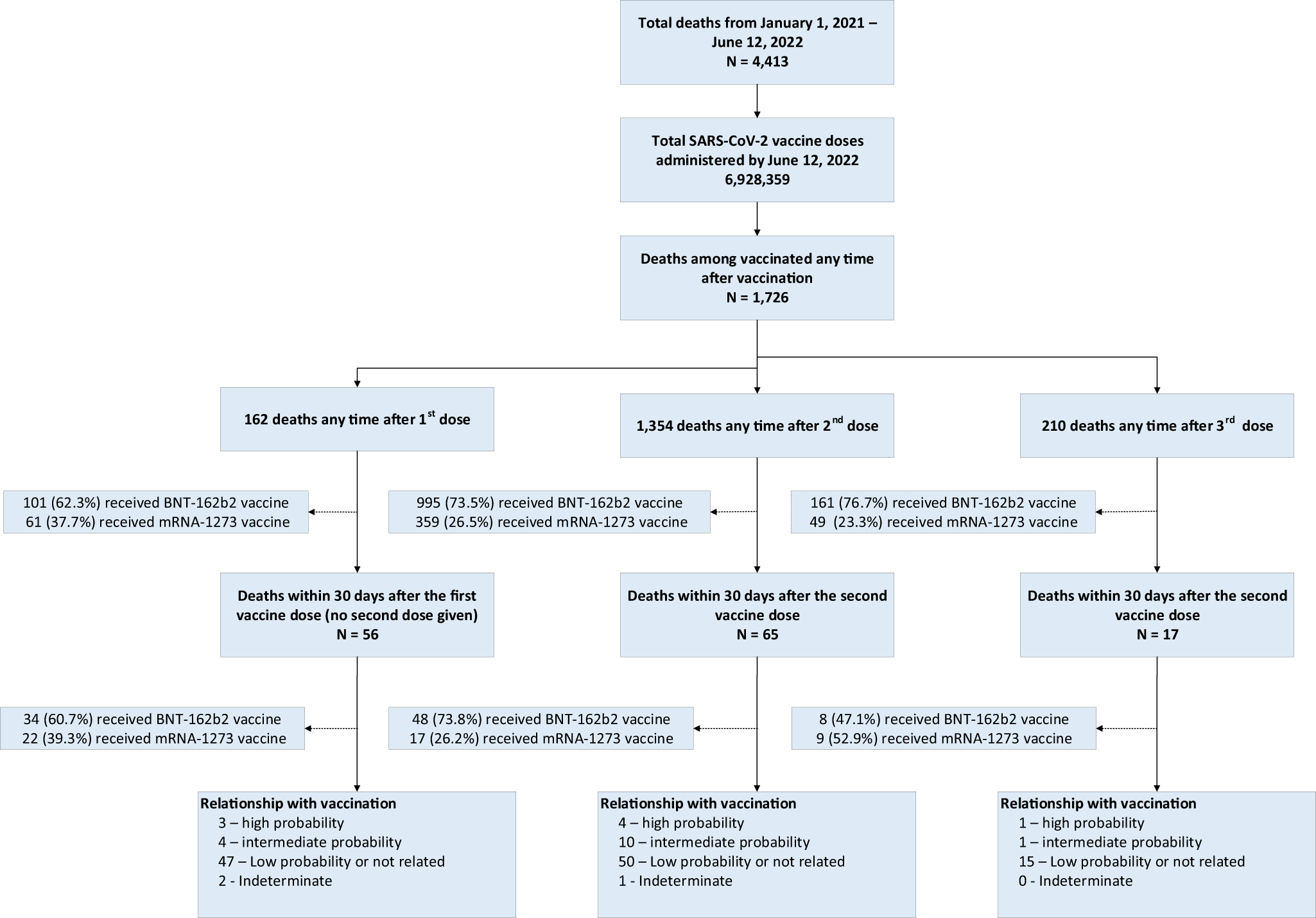
Evaluation of mortality attributable to SARS-CoV-2 vaccine administration using national level data from Qatar | Nature Communications

Qatar: Eligible Indian travellers, vaccinated with both does of Covidhield, can skip quarantine | Times of India Travel

Vaccine effectiveness of one, two, and three doses of BNT162b2 and CoronaVac against COVID-19 in Hong Kong: a population-based observational study - The Lancet Infectious Diseases

Effectiveness of a third dose of BNT162b2 mRNA COVID-19 vaccine in a large US health system: A retrospective cohort study - The Lancet Regional Health – Americas

Functional immune responses against SARS-CoV-2 variants of concern after fourth COVID-19 vaccine dose or infection in patients with blood cancer - ScienceDirect
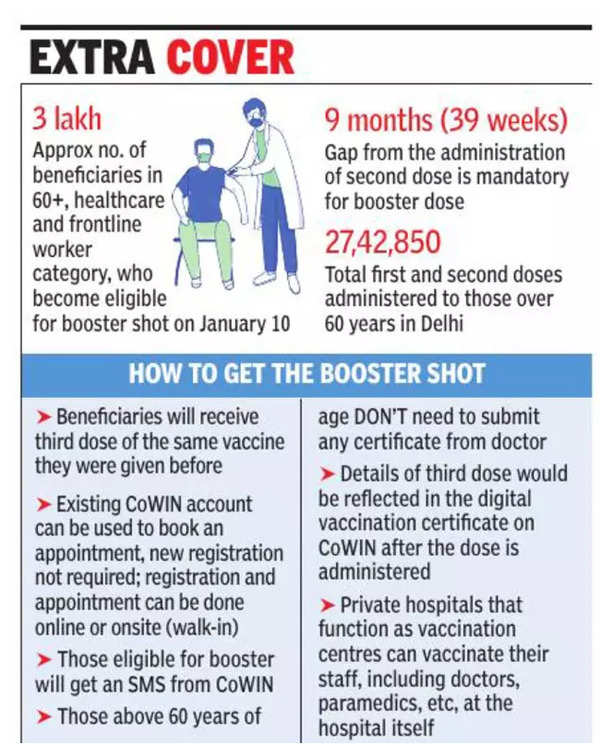
Delhi: Registration begins, vaccine booster shot for elderly from tomorrow | Delhi News - Times of India

Safety and Immunogenicity After a Three-Dose SARS-CoV-2 Vaccine Schedule in Allogeneic Stem Cell Transplant Recipients - Transplantation and Cellular Therapy, Official Publication of the American Society for Transplantation and Cellular Therapy

وزارة الصحة العامة on X: "The full quarantine exemption criteria are as follows: • 14 days must have passed after receiving the second dose of the vaccine before returning into Qatar and
Saudi Arabia Booster Dose Mandatory: Saudi Arabia makes booster shots mandatory for its citizens travelling abroad from February 9 | Times of India Travel

وزارة الصحة العامة on X: "Ministry of Public Health announces new Ehteraz functionalities for home quarantine and COVID-19 vaccination status.#YourSafetyIsMySafety https://t.co/tjZt2ya0r9" / X

Vaccines | Free Full-Text | Pfizer-BioNTech mRNA Vaccine Protection among Children and Adolescents Aged 12–17 Years against COVID-19 Infection in Qatar

Hamad Medical Corporation - COVID-19 vaccination certificate now available on MyHealth Patient Portal All individuals who have received 2 doses of the COVID-19 vaccine will automatically have a vaccination certificate available for

Safety and immunogenicity of seven COVID-19 vaccines as a third dose (booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -
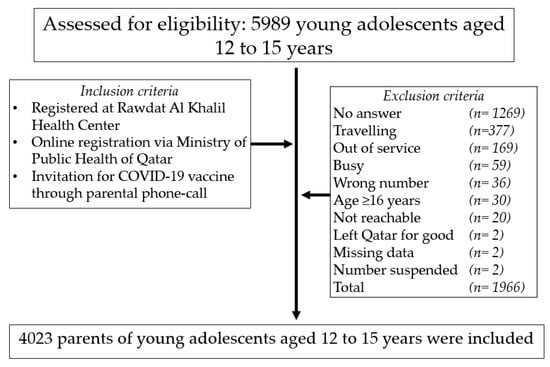
Vaccines | Free Full-Text | BNT162b2 COVID-19 Vaccine Hesitancy among Parents of 4023 Young Adolescents (12–15 Years) in Qatar