Moderate‐Affinity Affibodies Modulate the Delivery and Bioactivity of Bone Morphogenetic Protein‐2 - Dorogin - 2023 - Advanced Healthcare Materials - Wiley Online Library

Orotidine 5′-Monophosphate Decarboxylase: Probing the Limits of the Possible for Enzyme Catalysis | Accounts of Chemical Research

Toxins | Free Full-Text | Helicobacter pylori BabA–SabA Key Roles in the Adherence Phase: The Synergic Mechanism for Successful Colonization and Disease Development
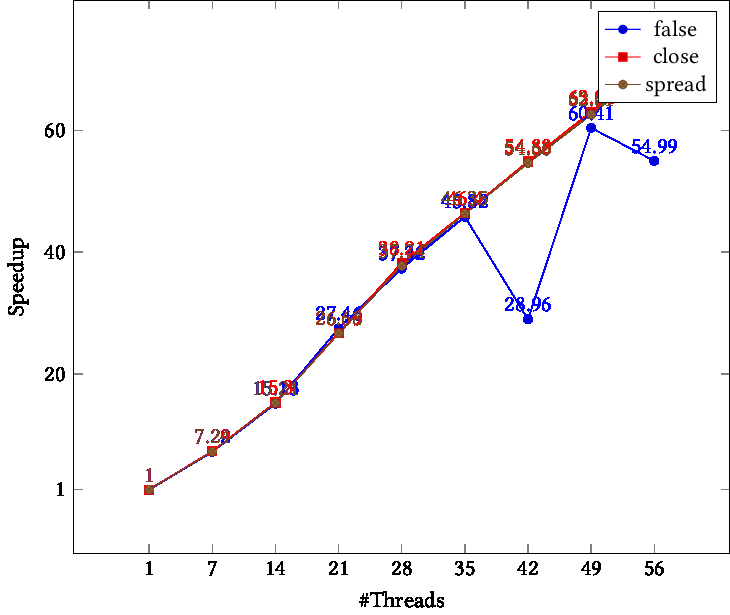
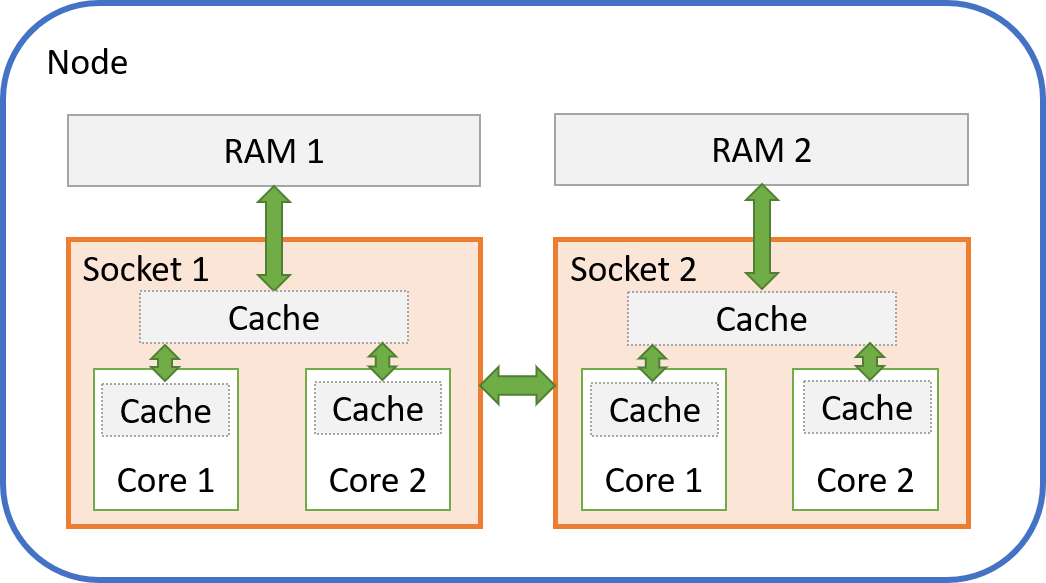
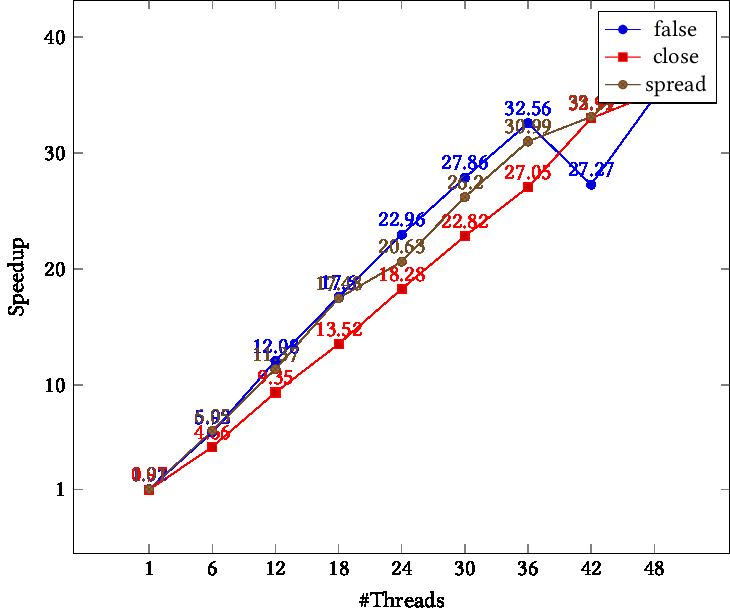
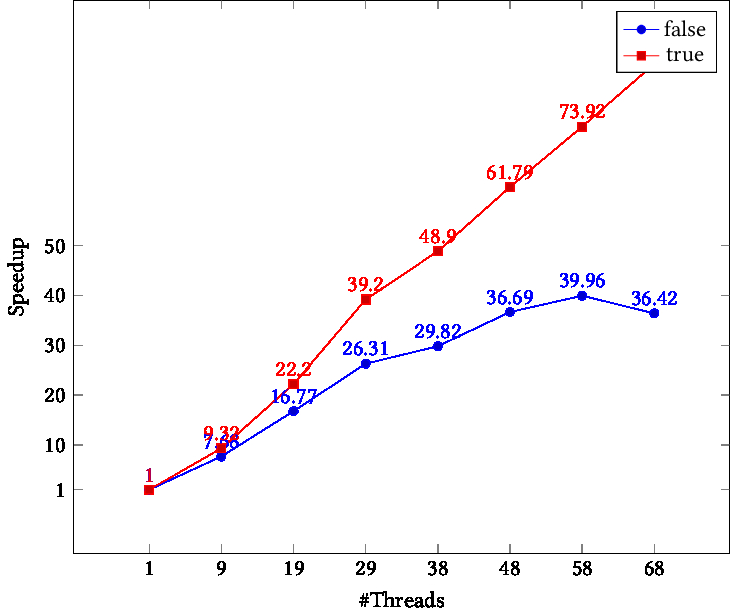
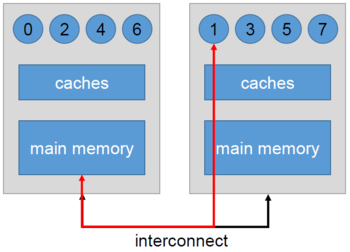
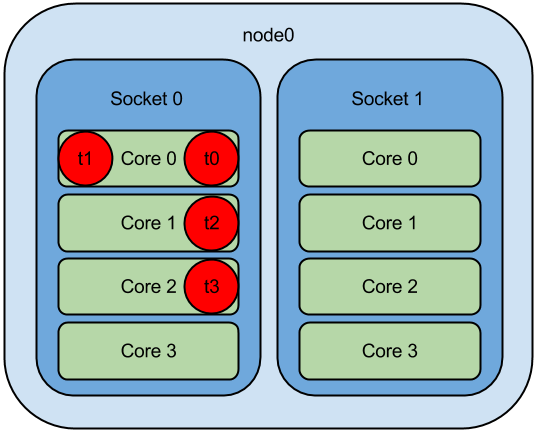
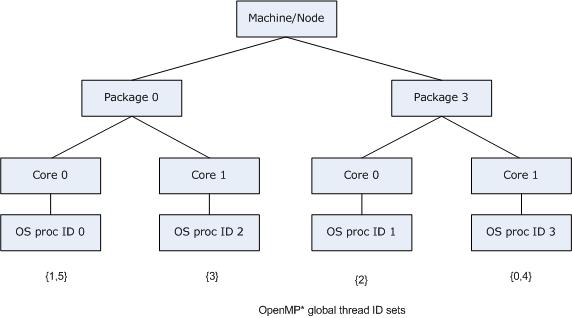
openblas runs single thread with OMP_PROC_BIND=TRUE or GOMP_CPU_AFFINITY · Issue #2238 · OpenMathLib/OpenBLAS · GitHub

Global Maps of ProQ Binding In Vivo Reveal Target Recognition via RNA Structure and Stability Control at mRNA 3′ Ends - ScienceDirect
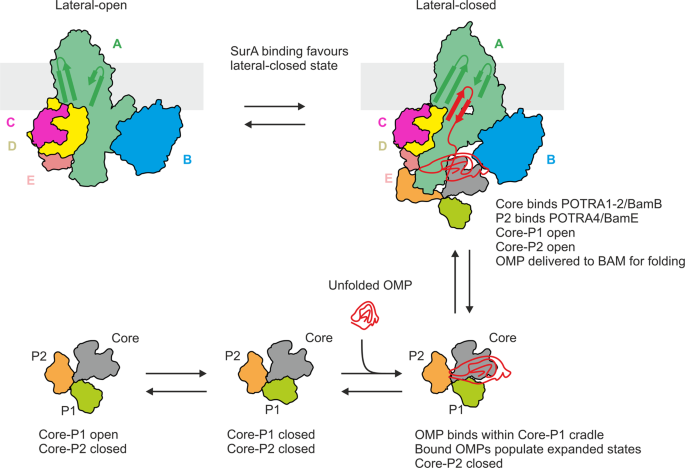
Dynamic interplay between the periplasmic chaperone SurA and the BAM complex in outer membrane protein folding | Communications Biology

Utilization of Cofactor Binding Energy for Enzyme Catalysis: Formate Dehydrogenase-Catalyzed Reactions of the Whole NAD Cofactor and Cofactor Pieces | Biochemistry

RNA-binding activity and regulatory functions of the emerging sRNA-binding protein ProQ - ScienceDirect

Inter-domain dynamics in the chaperone SurA and multi-site binding to its unfolded outer membrane protein clients | bioRxiv