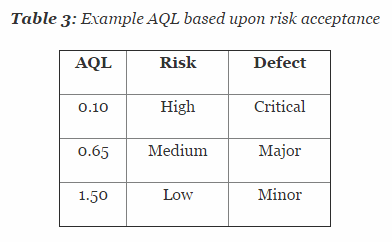
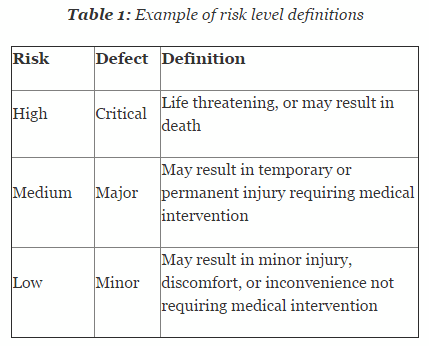
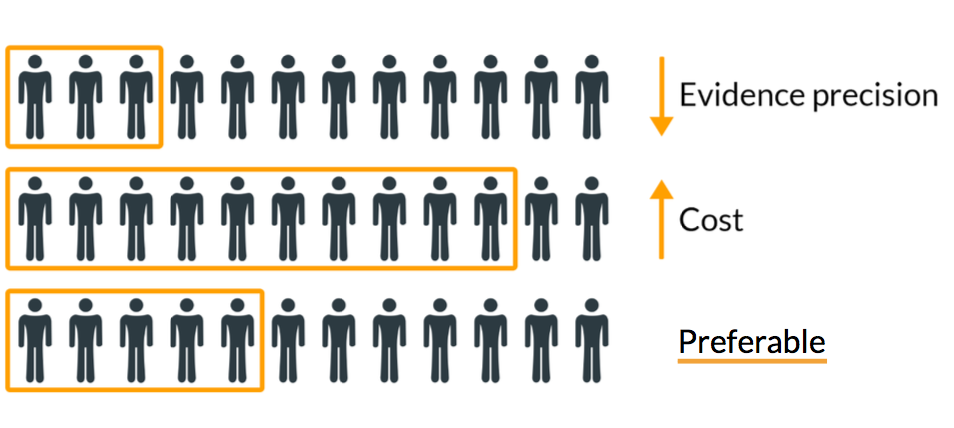
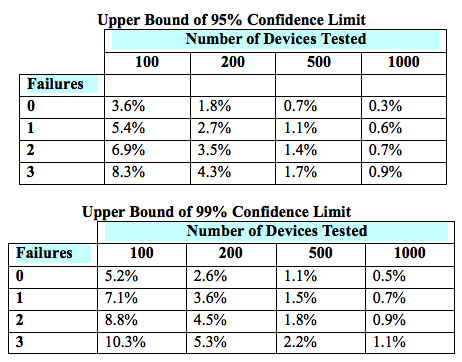
Applications for Medical Device Investigational Testing Authorizations Guidance Document - Canada.ca
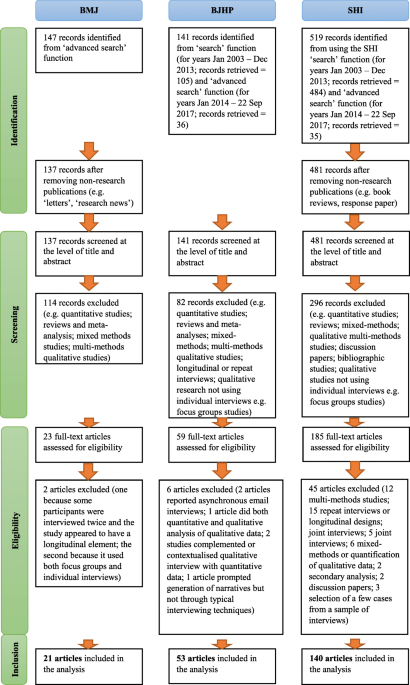
Characterising and justifying sample size sufficiency in interview-based studies: systematic analysis of qualitative health research over a 15-year period | BMC Medical Research Methodology | Full Text

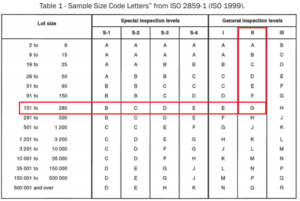
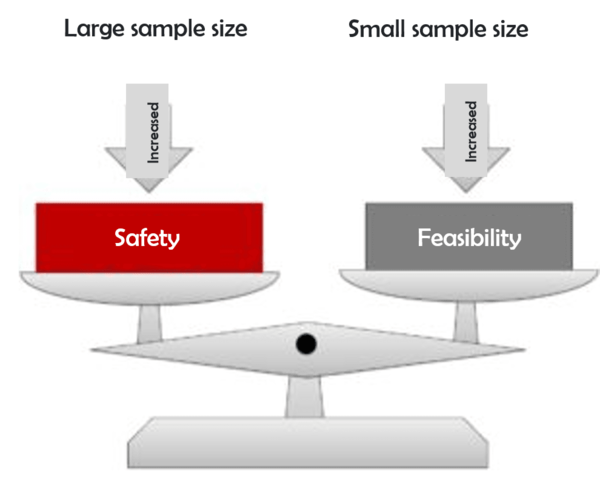
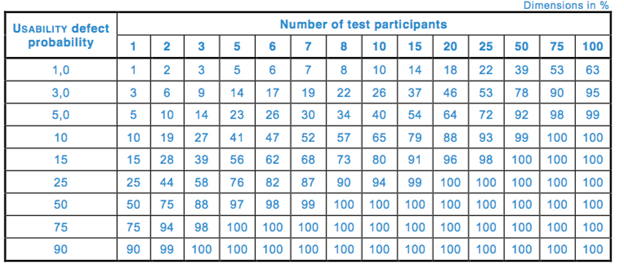
![ARTICLE] Sampling Strategy: 10 FAQs on Sample Size Selection - Medidee Services ARTICLE] Sampling Strategy: 10 FAQs on Sample Size Selection - Medidee Services](https://medidee.com/wp-content/uploads/2022/08/Mdd-Blog-Posts-Images-1-1.png)




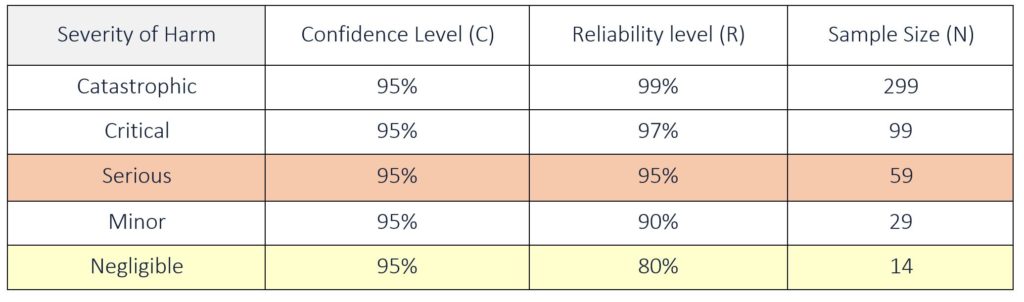


![ARTICLE] Sampling Strategy: 10 FAQs on Sample Size Selection - Medidee Services ARTICLE] Sampling Strategy: 10 FAQs on Sample Size Selection - Medidee Services](https://medidee.com/wp-content/uploads/2022/08/Technical-Documentation-Infographic.png)