Passenger sequences can promote interlaced dimers in a common variant of the maltose-binding protein | Scientific Reports

Maltose-binding Protein Is Open in the Catalytic Transition State for ATP Hydrolysis during Maltose Transport - ScienceDirect

Positional effects of fusion partners on the yield and solubility of MBP fusion proteins - ScienceDirect
Expression of proteins in Escherichia coli as fusions with maltose-binding protein to rescue non-expressed targets in a high-thr
Maltose-Binding Protein (MBP), a Secretion-Enhancing Tag for Mammalian Protein Expression Systems | PLOS ONE

Purified human mini-calpain-5 is well folded (A) The maltose binding... | Download Scientific Diagram

A peptide/maltose-binding protein fusion protein used to replace the traditional antigen for immunological detection of deoxynivalenol in food and feed - ScienceDirect
Maltose-Binding Protein Fusion Allows for High Level Bacterial Expression and Purification of Bioactive Mammalian Cytokine Derivatives | PLOS ONE

Systematic analysis of the expression, solubility and purification of a passenger protein in fusion with different tags - ScienceDirect

Passenger sequences can promote interlaced dimers in a common variant of the maltose-binding protein | Scientific Reports
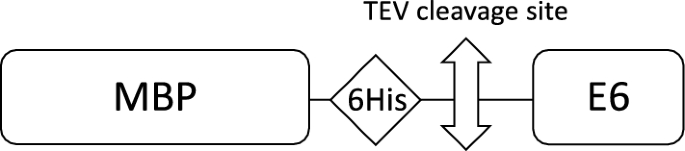
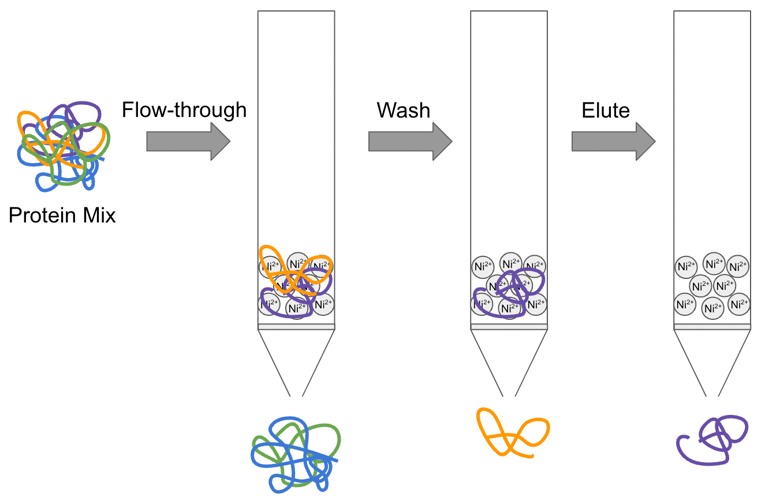
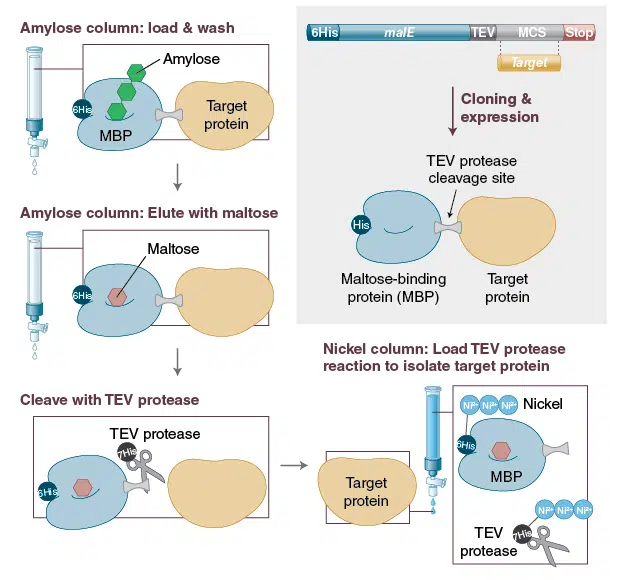
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text

A) Schematic representation of the pLic-MBP expression vector using... | Download Scientific Diagram

High-yield soluble expression, purification and characterization of human steroidogenic acute regulatory protein (StAR) fused to a cleavable Maltose-Binding Protein (MBP) - ScienceDirect

Alignment of deduced amino acid sequences for maltose-binding proteins.... | Download Scientific Diagram