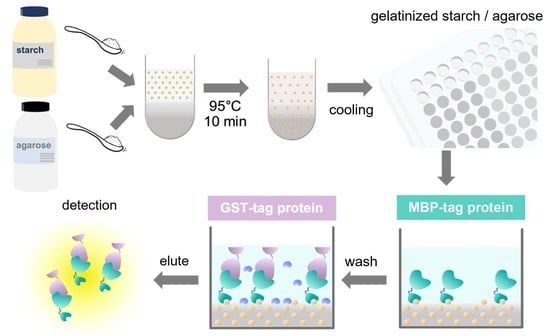
MPs | Free Full-Text | A Cost-Effective Immobilization Method for MBP Fusion Proteins on Microtiter Plates Using a Gelatinized Starch–Agarose Mixture and Its Application for Convenient Protein–Protein Interaction Analysis
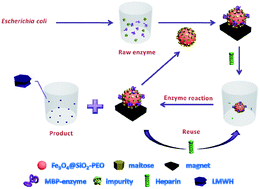
Magnetic nanoparticles for the affinity adsorption of maltose binding protein (MBP) fusion enzymes - Journal of Materials Chemistry (RSC Publishing)
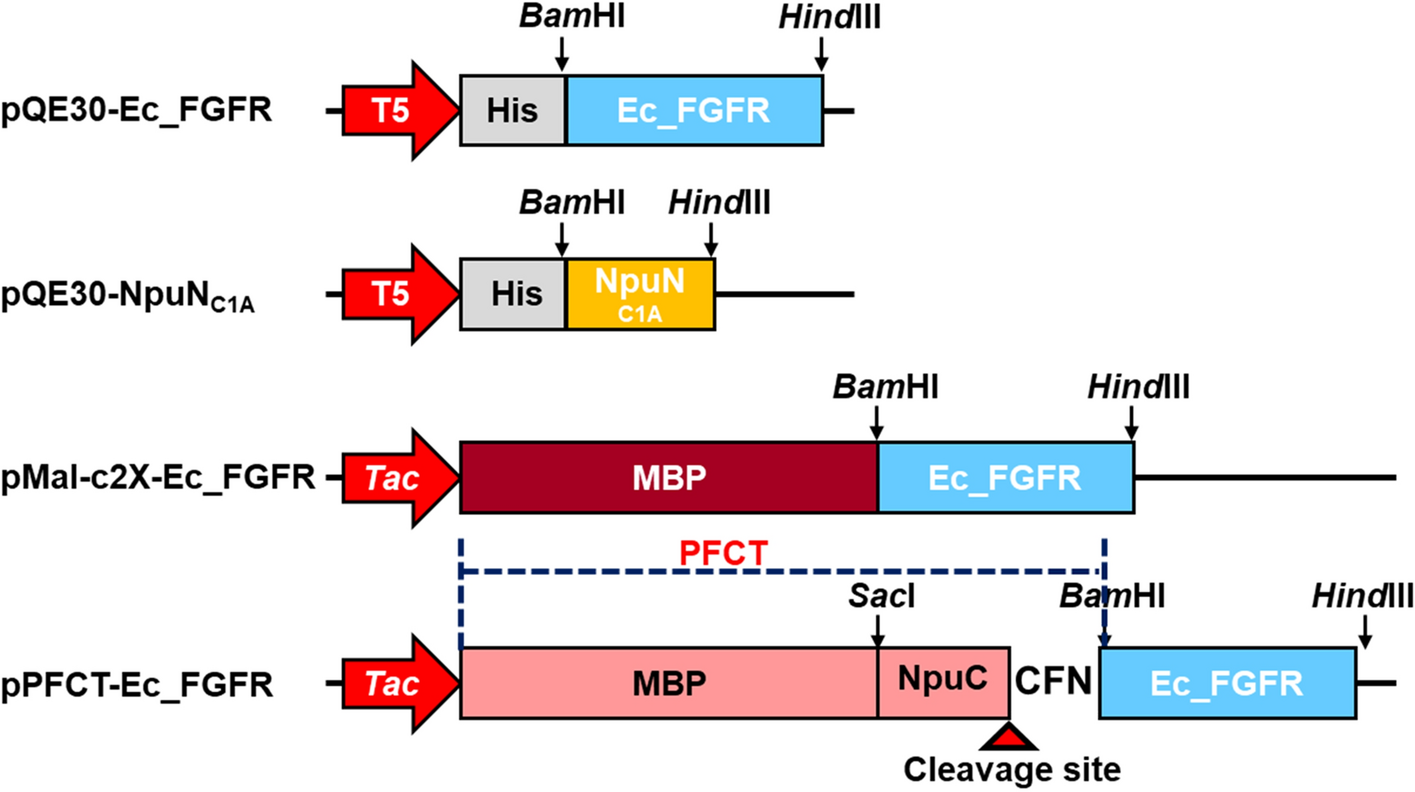
A designed fusion tag for soluble expression and selective separation of extracellular domains of fibroblast growth factor receptors | Scientific Reports

Evaluation of Maltose Binding Protein-Tagged hATR Kinase Domain Catalytic Activity with p53 Ser-15 Phosphorylation | Biochemistry
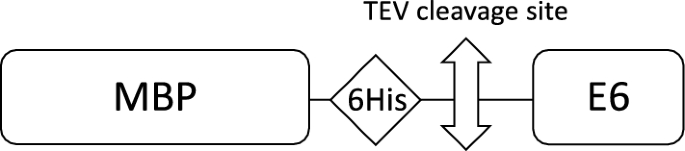
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text
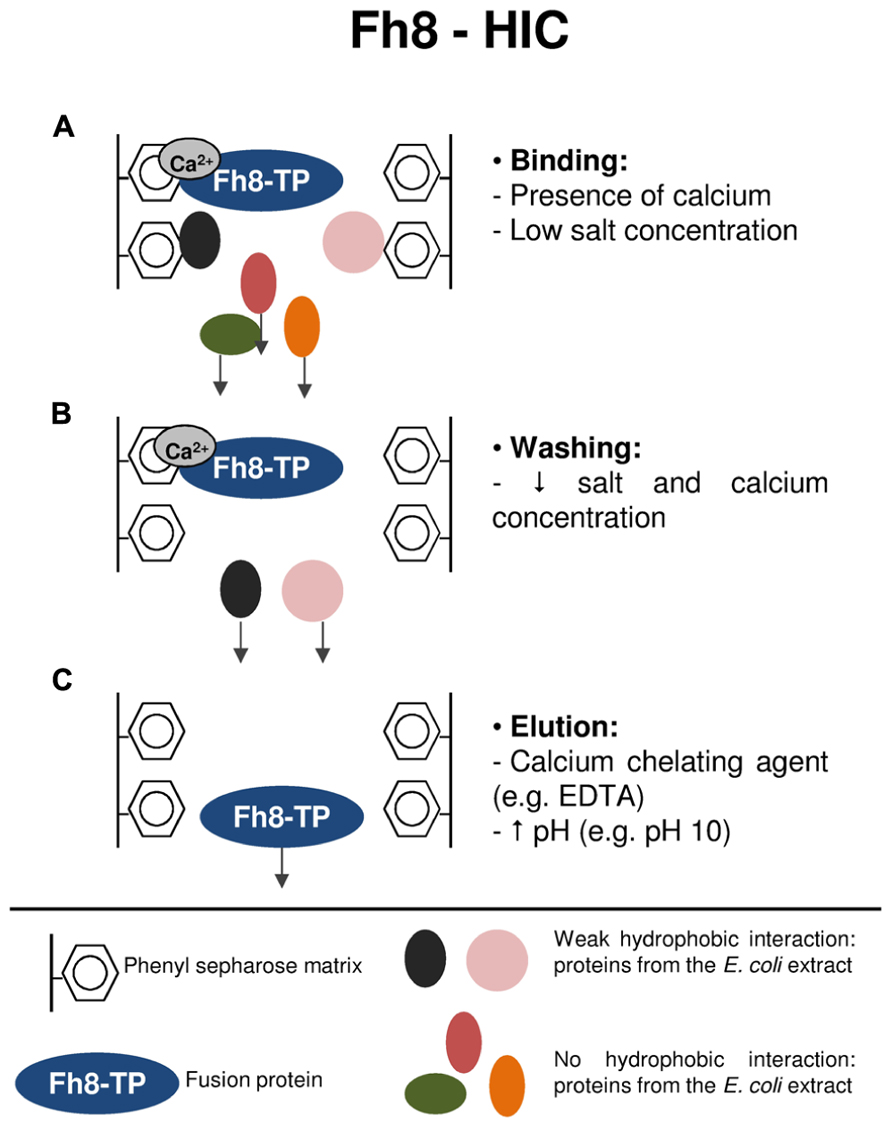
Frontiers | Fusion tags for protein solubility, purification and immunogenicity in Escherichia coli: the novel Fh8 system

Purification of fibroblast growth factor 1 (FGF1) by receptor affinity... | Download Scientific Diagram
The Ability to Enhance the Solubility of Its Fusion Partners Is an Intrinsic Property of Maltose-Binding Protein but Their Folding Is Either Spontaneous or Chaperone-Mediated | PLOS ONE

Mutations in maltose-binding protein that alter affinity and solubility properties | Applied Microbiology and Biotechnology

Prokaryotic soluble expression and purification of bioactive human fibroblast growth factor 21 using maltose-binding protein | Scientific Reports