![Electrospray ionization photoelectron spectroscopy of cryogenic [EDTA·M(ii)]2− complexes (M = Ca, V–Zn): electronic structures and intrinsic redox properties - Faraday Discussions (RSC Publishing) Electrospray ionization photoelectron spectroscopy of cryogenic [EDTA·M(ii)]2− complexes (M = Ca, V–Zn): electronic structures and intrinsic redox properties - Faraday Discussions (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C8FD00175H)
Electrospray ionization photoelectron spectroscopy of cryogenic [EDTA·M(ii)]2− complexes (M = Ca, V–Zn): electronic structures and intrinsic redox properties - Faraday Discussions (RSC Publishing)
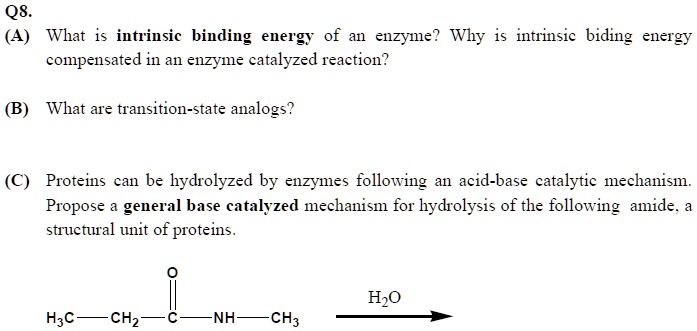
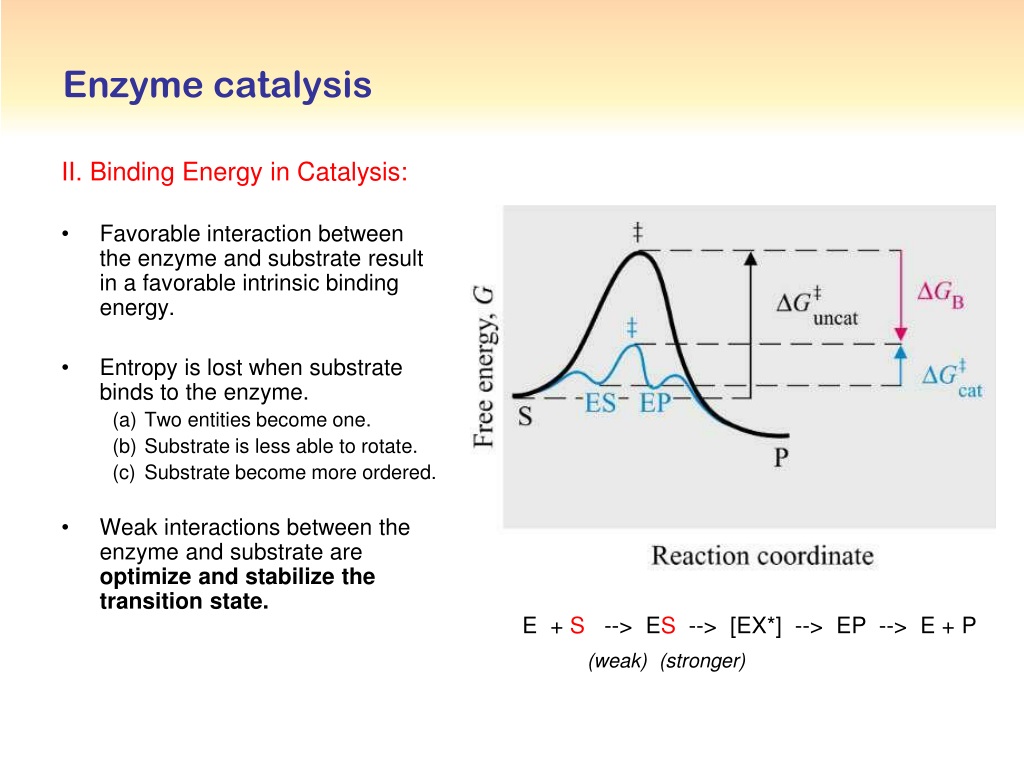
SOLVED: Q8 (4) What is the intrinsic binding energy of an enzyme? Why is it compensated in an enzyme-catalyzed reaction? Intrinsic binding energy What are transition-state analogs? Proteins can be hydrolyzed by

A) Partitioning of the intrinsic transition state binding energy of... | Download Scientific Diagram

Full article: Intrinsic thermodynamics of sulfonamide inhibitor binding to human carbonic anhydrases I and II
![PDF] Utilization of Substrate Intrinsic Binding Energy for Conformational Change and Catalytic Function in Phosphoenolpyruvate Carboxykinase. | Semantic Scholar PDF] Utilization of Substrate Intrinsic Binding Energy for Conformational Change and Catalytic Function in Phosphoenolpyruvate Carboxykinase. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/faf53247a304d285742e04a0ac39592a976bbbfe/3-Table1-1.png)
PDF] Utilization of Substrate Intrinsic Binding Energy for Conformational Change and Catalytic Function in Phosphoenolpyruvate Carboxykinase. | Semantic Scholar

Binding Energy and Free Energy of Calcium Ion to Calmodulin EF-Hands with the Drude Polarizable Force Field | ACS Physical Chemistry Au
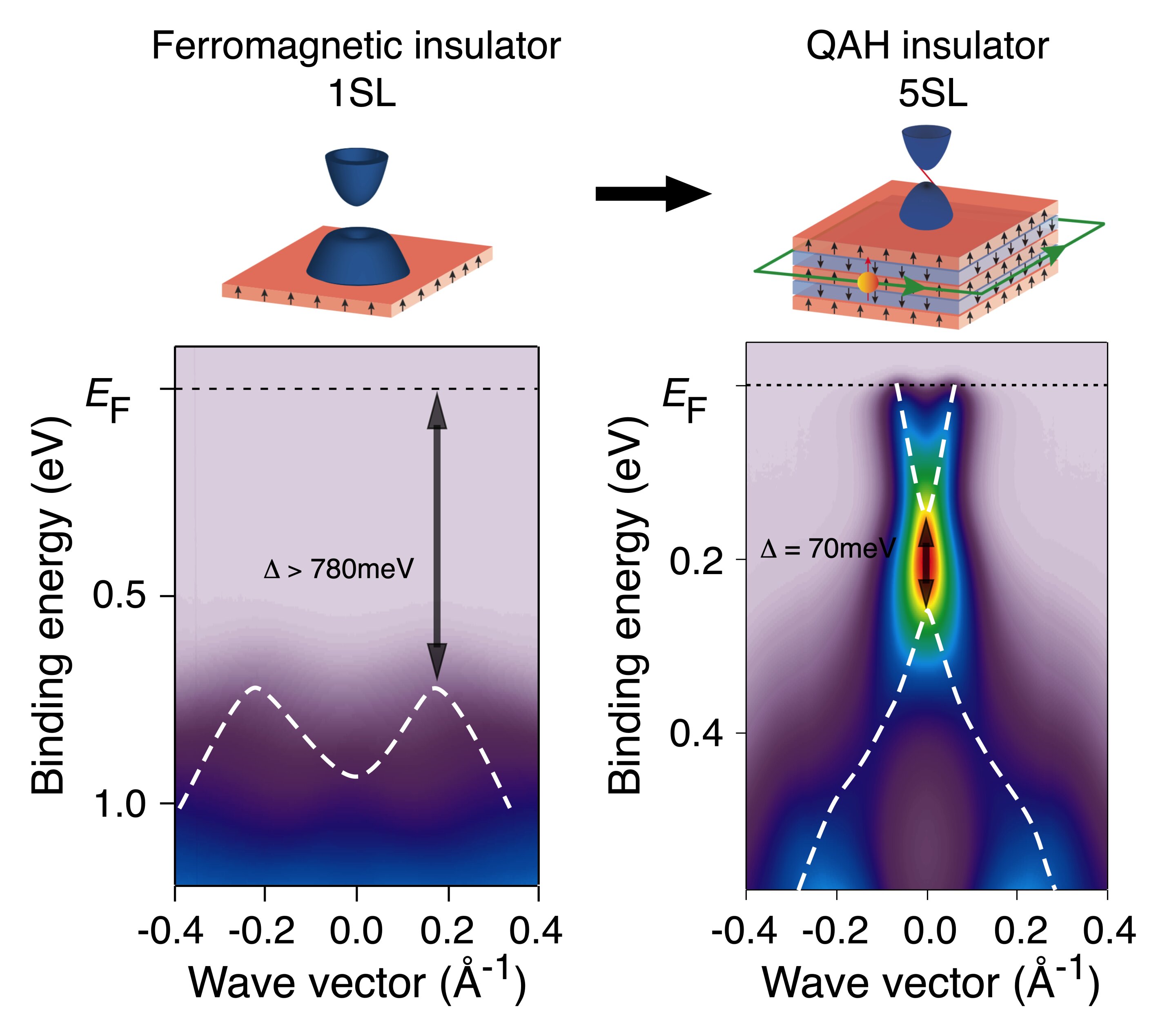
![PDF] Realizing an intrinsic excitonic insulator by decoupling exciton binding energy from the minimum band gap | Semantic Scholar PDF] Realizing an intrinsic excitonic insulator by decoupling exciton binding energy from the minimum band gap | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/750e9e4d5276c6d505c9a18e45667cf73f057212/4-Figure1-1.png)
PDF] Realizing an intrinsic excitonic insulator by decoupling exciton binding energy from the minimum band gap | Semantic Scholar
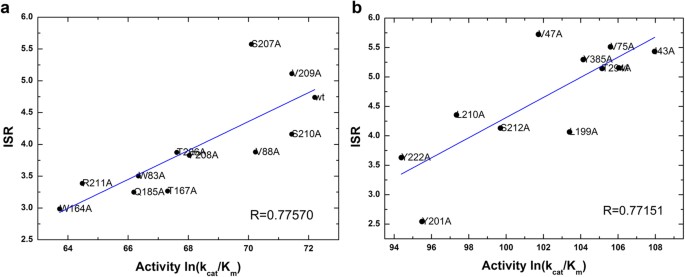
Energy Landscape Topography Reveals the Underlying Link Between Binding Specificity and Activity of Enzymes | Scientific Reports