Effectiveness of less than three doses of quadrivalent human papillomavirus vaccine against cervical intraepithelial neoplasia when administered using a standard dose spacing schedule: Observational cohort of young women in Australia - ScienceDirect

Single-dose HPV vaccination efficacy among adolescent girls and young women in Kenya (the KEN SHE Study): study protocol for a randomized controlled trial | Trials | Full Text

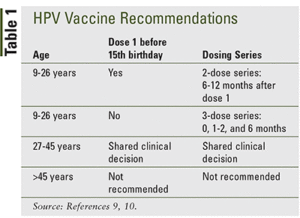
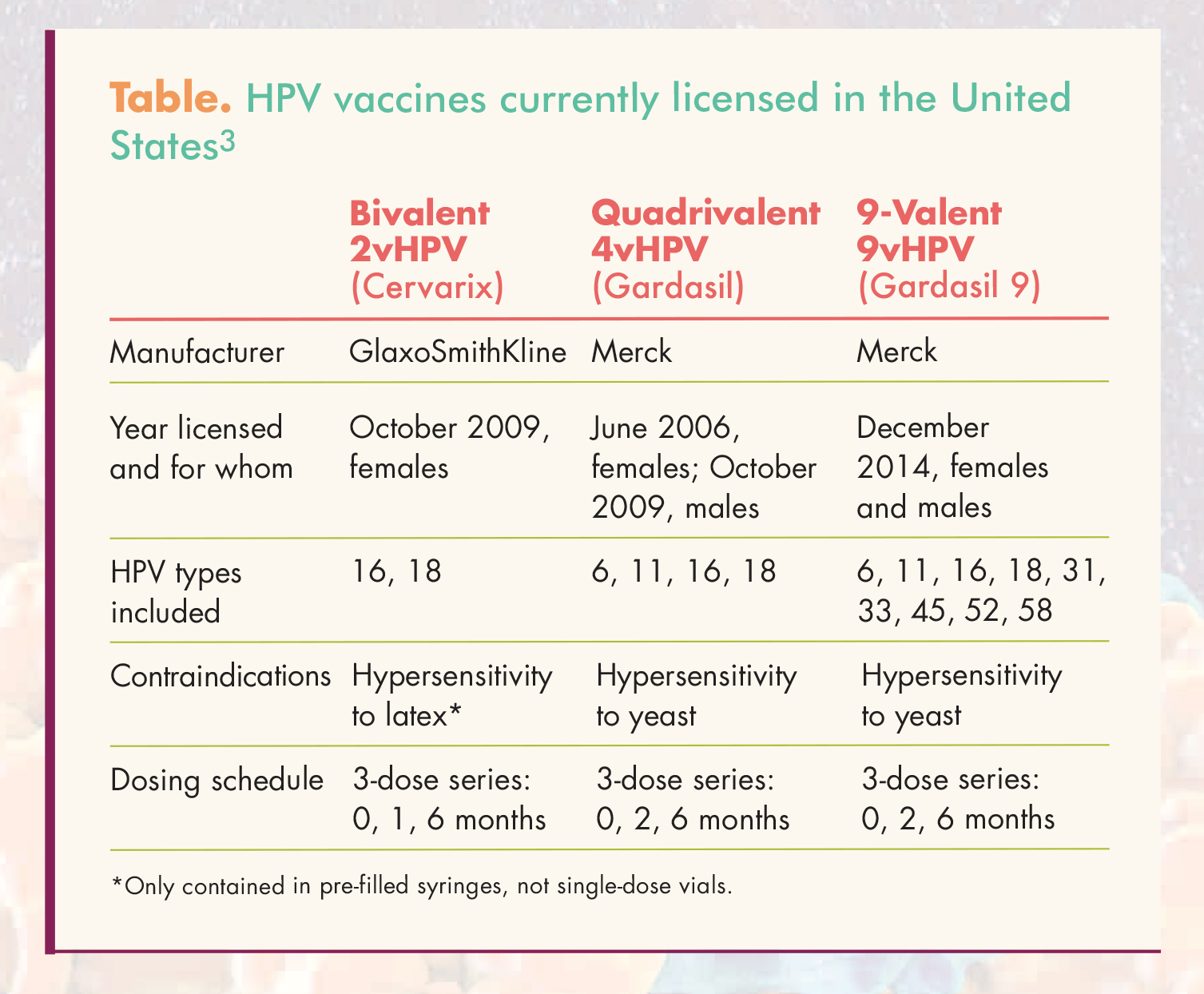
Updated Recommendations on Human Papillomavirus Vaccines: 9-valent HPV vaccine and clarification of minimum intervals between doses in the HPV immunization schedule - Canada.ca

Uptake of HPV vaccination among boys after the introduction of gender-neutral HPV vaccination in Germany before and during the COVID-19 pandemic | Infection

The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study - The Lancet

Milestones and timelines in the late stage clinical development and... | Download Scientific Diagram

Updated Recommendations on Human Papillomavirus Vaccines: 9-valent HPV vaccine and clarification of minimum intervals between doses in the HPV immunization schedule - Canada.ca

Effects of the age of vaccination on the humoral responses to a human papillomavirus vaccine | npj Vaccines

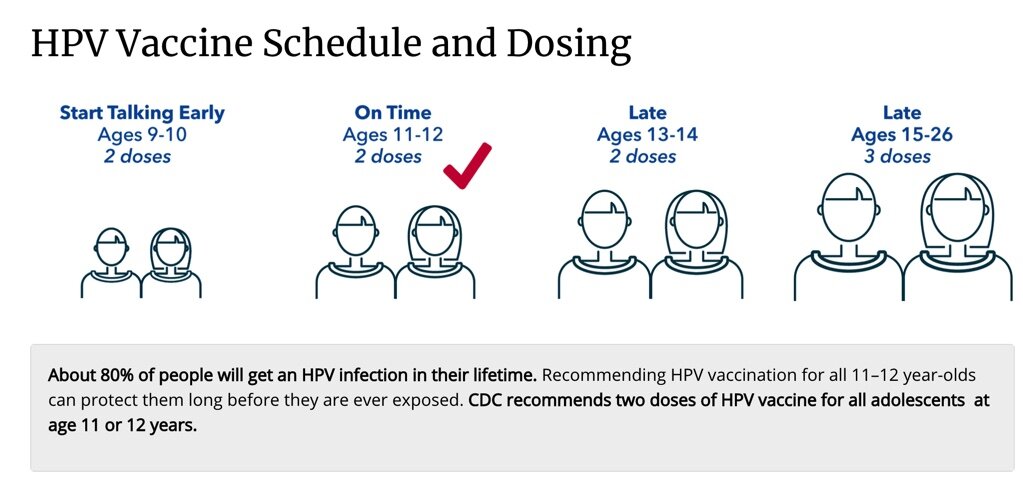
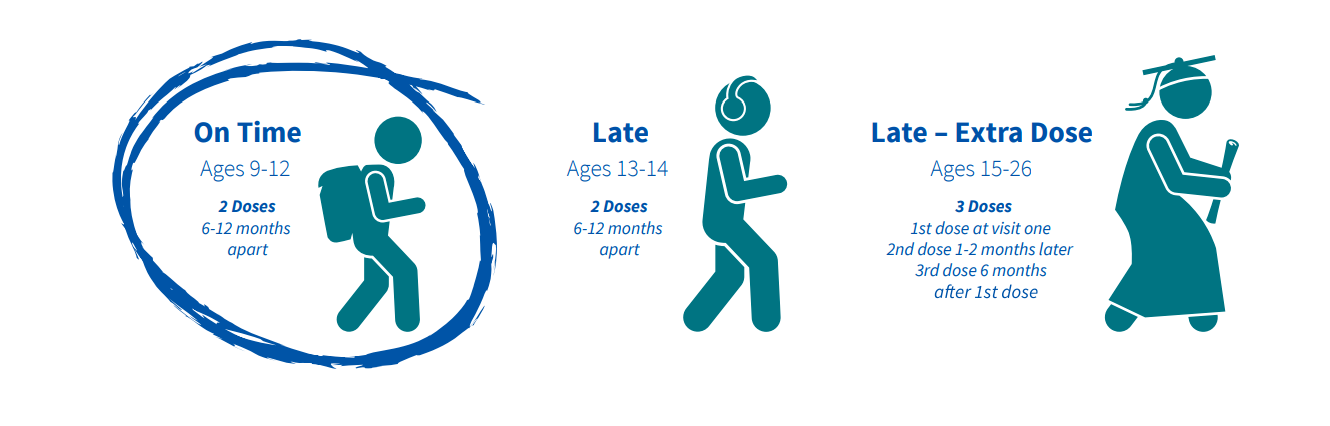
Vax 2 Stop Cancer on X: "The HPV vaccine is recommended for everyone ages 9–26. The ideal time for vaccination is between ages 9–12, with completion by the 13th birthday. Those ages