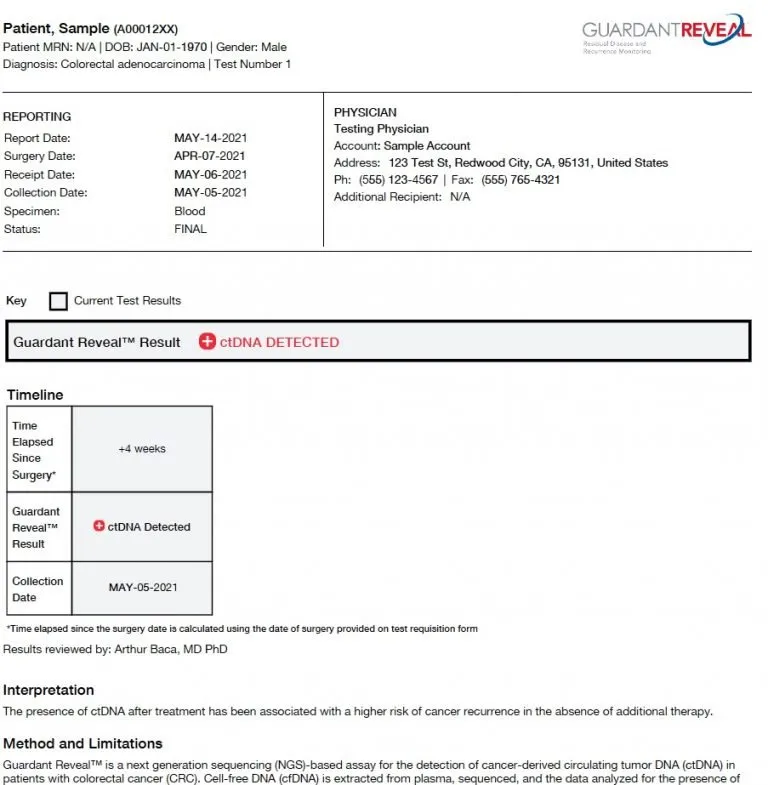
Analytical and Clinical Validation of a Digital Sequencing Panel for Quantitative, Highly Accurate Evaluation of Cell-Free Circulating Tumor DNA | PLOS ONE

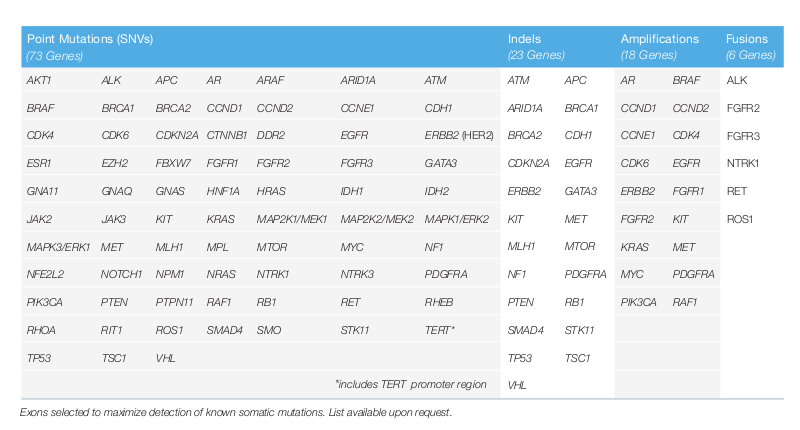
Test your advanced stage cancer patients with Guardant360®, get results in approximately 7 days - Guardant Health AMEA

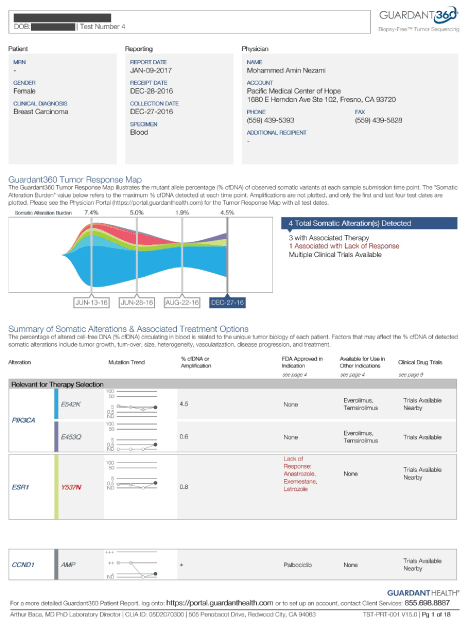
Role of Anti Angiogenic Therapy in Prevention of Recurrence in Hormonal Positive Breast Cancer: A Secondary Prevention Strategy and Method of Therapy

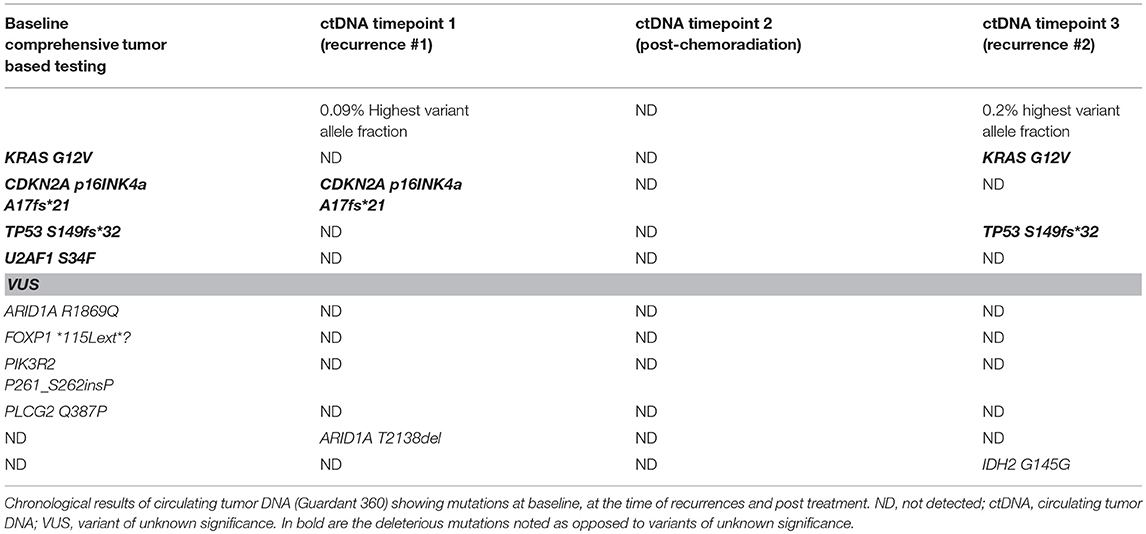
Circulating tumor DNA dynamics in advanced breast cancer treated with CDK4/6 inhibition and endocrine therapy | npj Breast Cancer