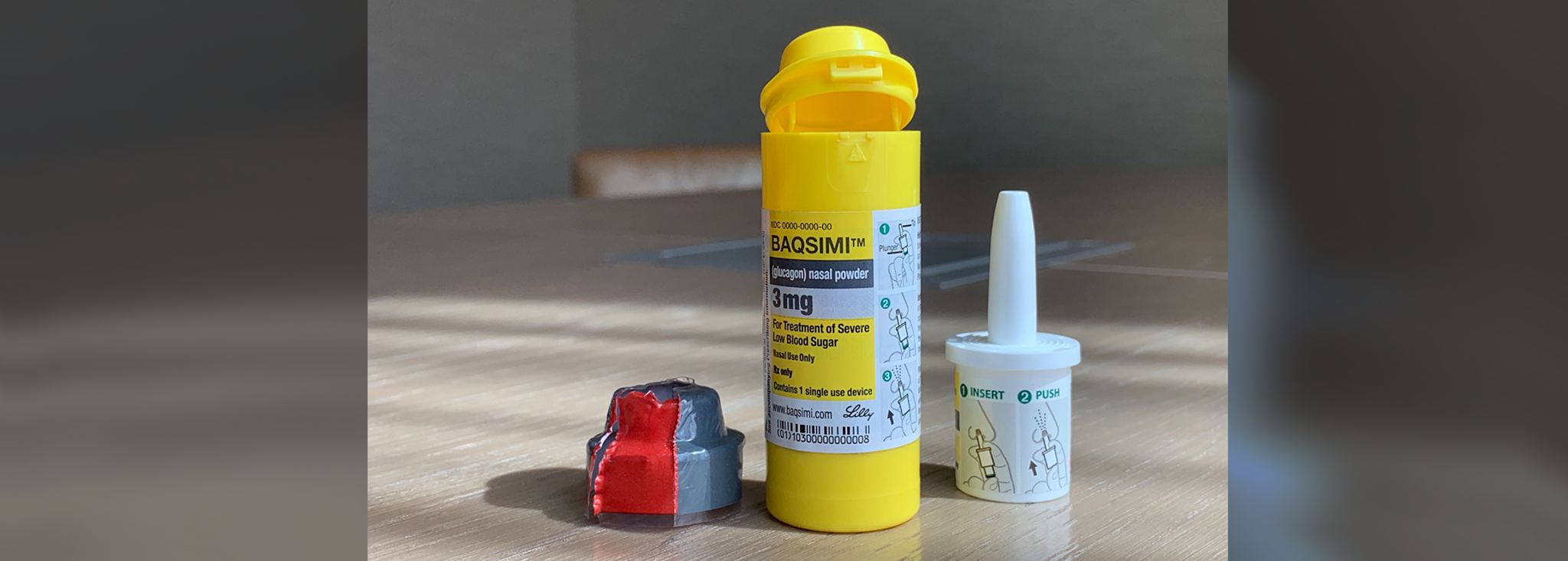
FDA Approves First Treatment for Severe Hypoglycemia That Can Be Administered without an Injection - Endocrine News

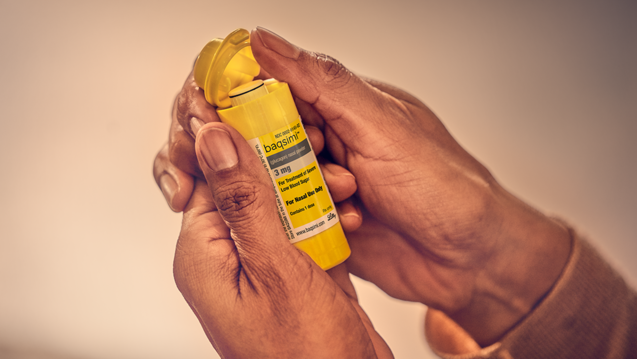
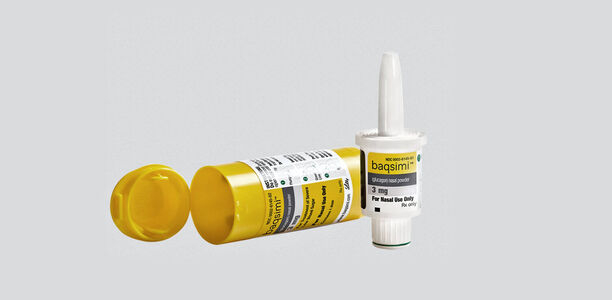
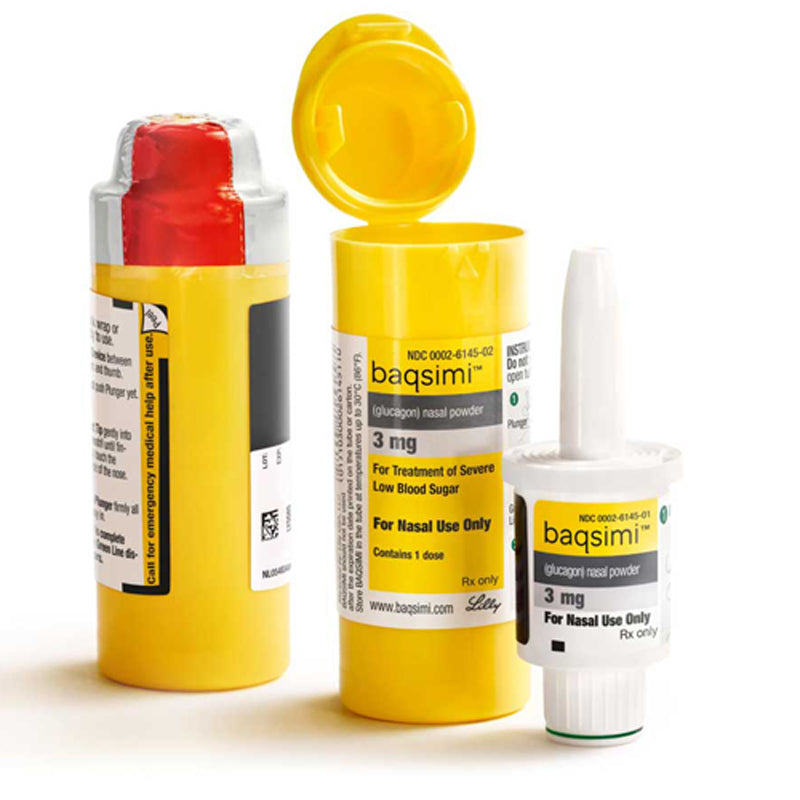
News Today Channel - The FDA has approved Baqsimi, a nasally administered glucagon, for treatment of severe hypoglycemia in people with diabetes four years & older. It is administered like a nasal

Temperaturwechsel kein Problem: Nasales Glucagon-Notfallpräparat erfüllt seine Aufgabe sowohl bei Hitze als auch Kälte

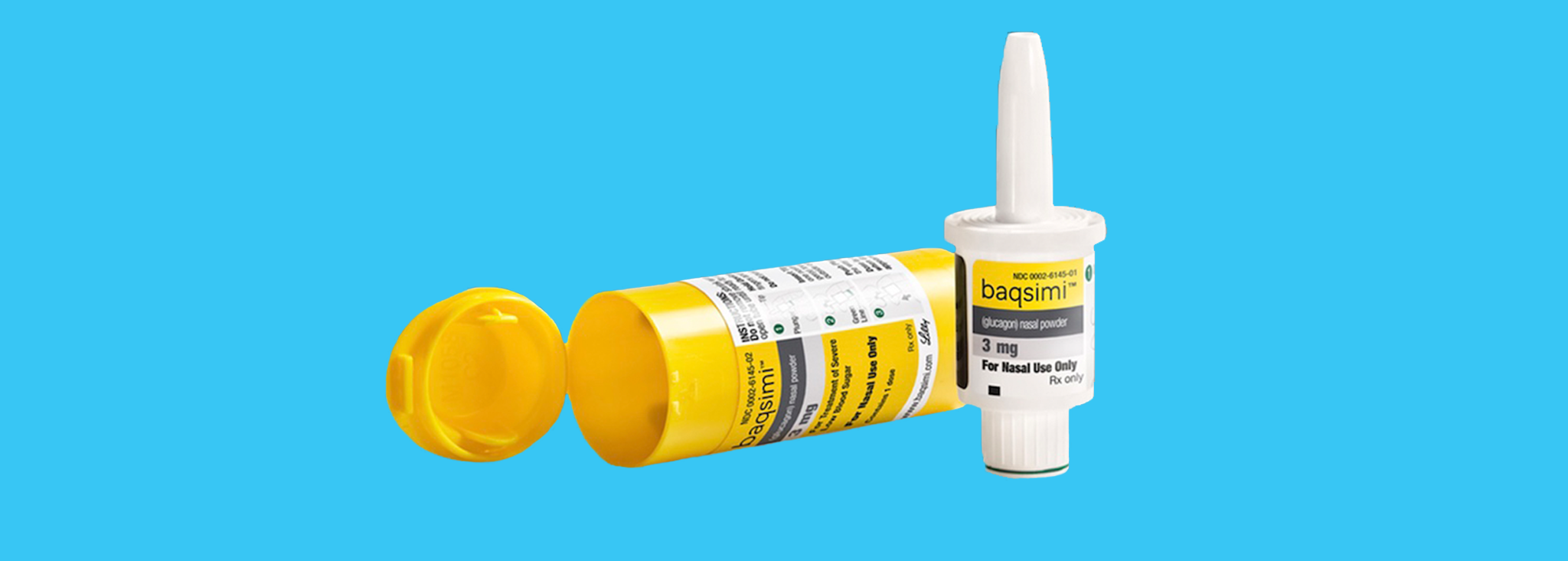
New nasal spray launched in Canada to combat hypoglycemic shock in diabetics - Greater Victoria News