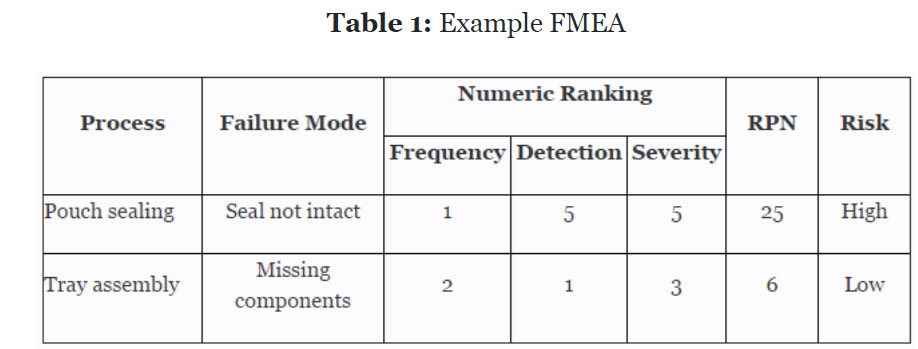
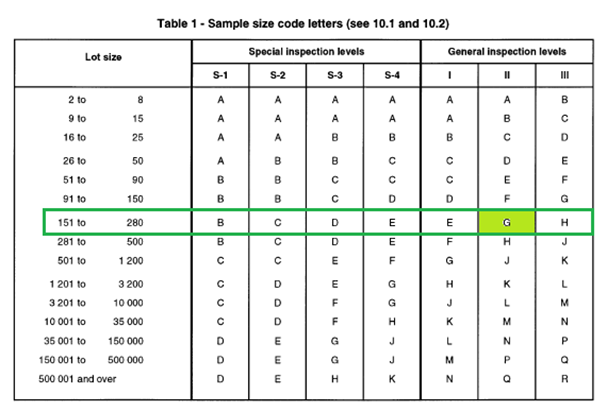
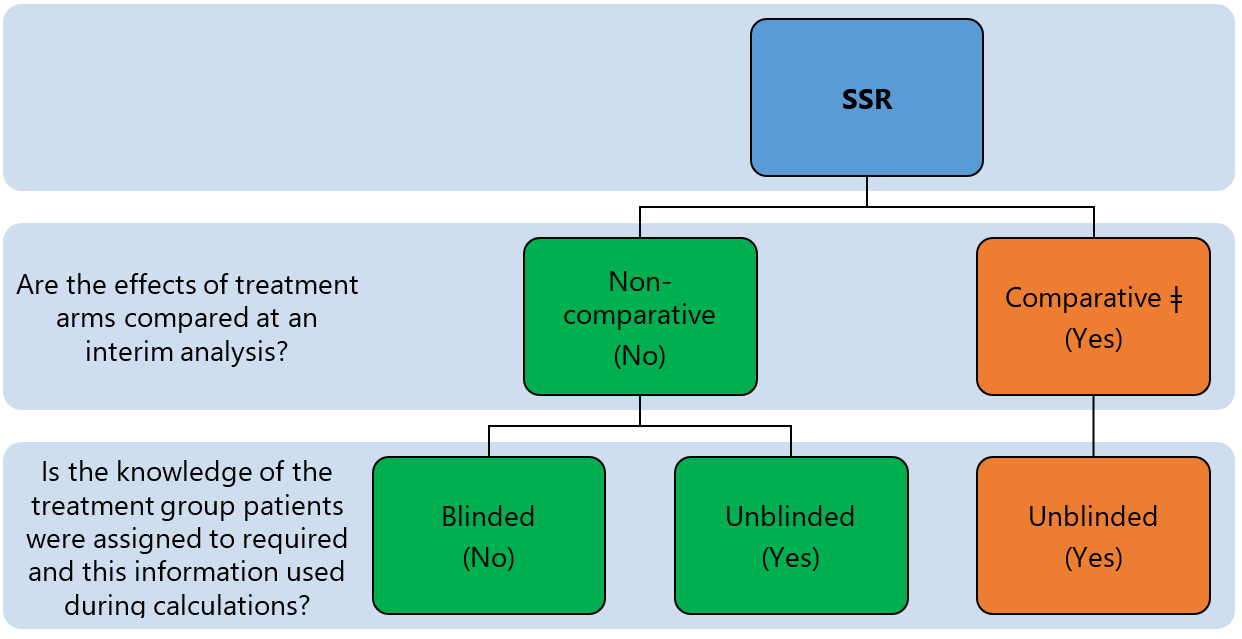
Recent innovations in adaptive trial designs: A review of design opportunities in translational research | Journal of Clinical and Translational Science | Cambridge Core

Recommendations for the Assessment of Blend and Content Uniformity: Modifications to Withdrawn FDA Draft Stratified Sampling Guidance | Journal of Pharmaceutical Innovation

Adaptive sample size reestimation in east infosheet by James. [email protected] - Issuu

Magnitude of effect and sample size justification in trials supporting anti-cancer drug approval by the US Food and Drug Administration | Scientific Reports

Early Feasibility Studies for Cardiovascular Devices in the United States: JACC State-of-the-Art Review - ScienceDirect