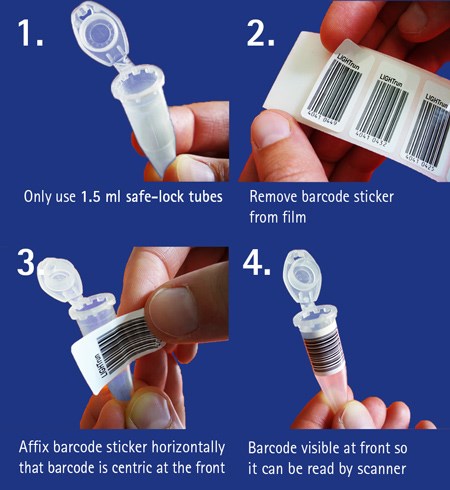
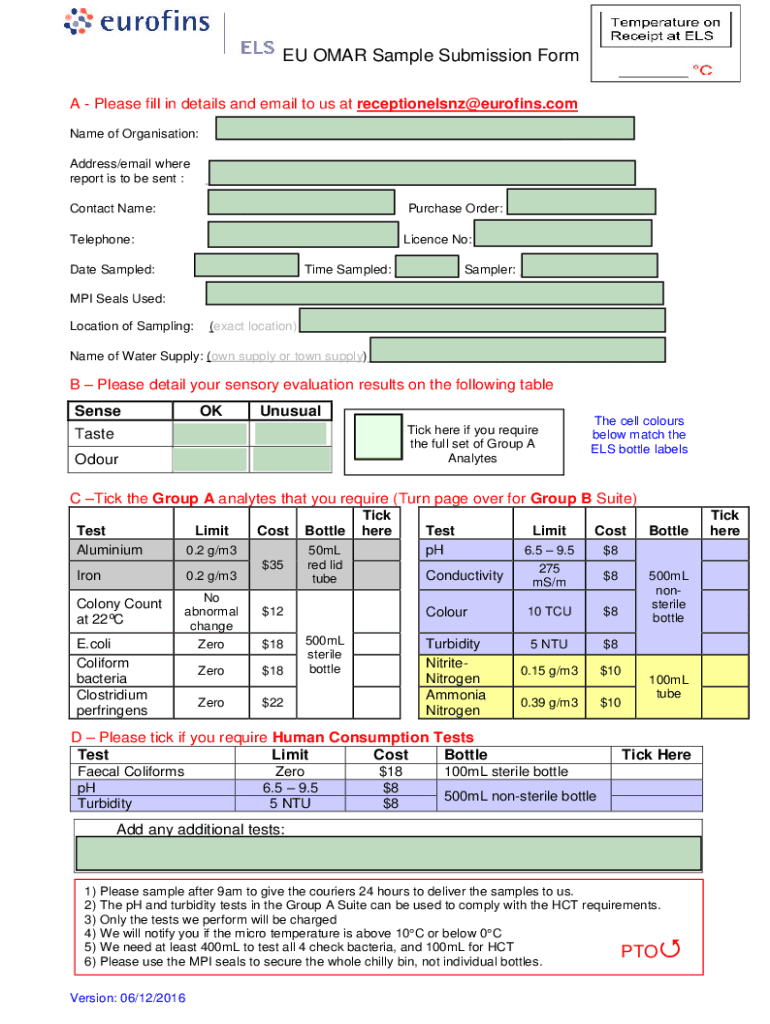
BIOBURDEN TESTING SAMPLE SUBMISSION FORM (DEVICES & PROCEDURAL PACKS) Client Information Company Name: Purchase Order (if av

Chemical Characterization: Current Status of Recent Changes and Some Interpretations Confirmation - Eurofins Deutschland

Biological Safety Spotlight: Surface-contacting Medical Devices. Current requirements and pitfalls explained using case studies. - Eurofins Deutschland