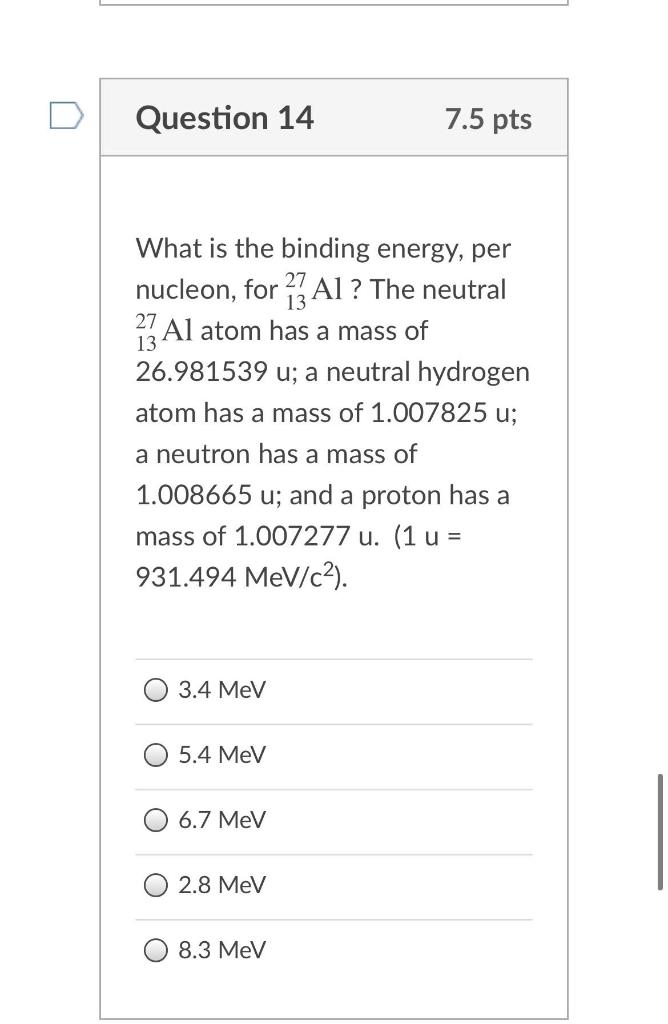
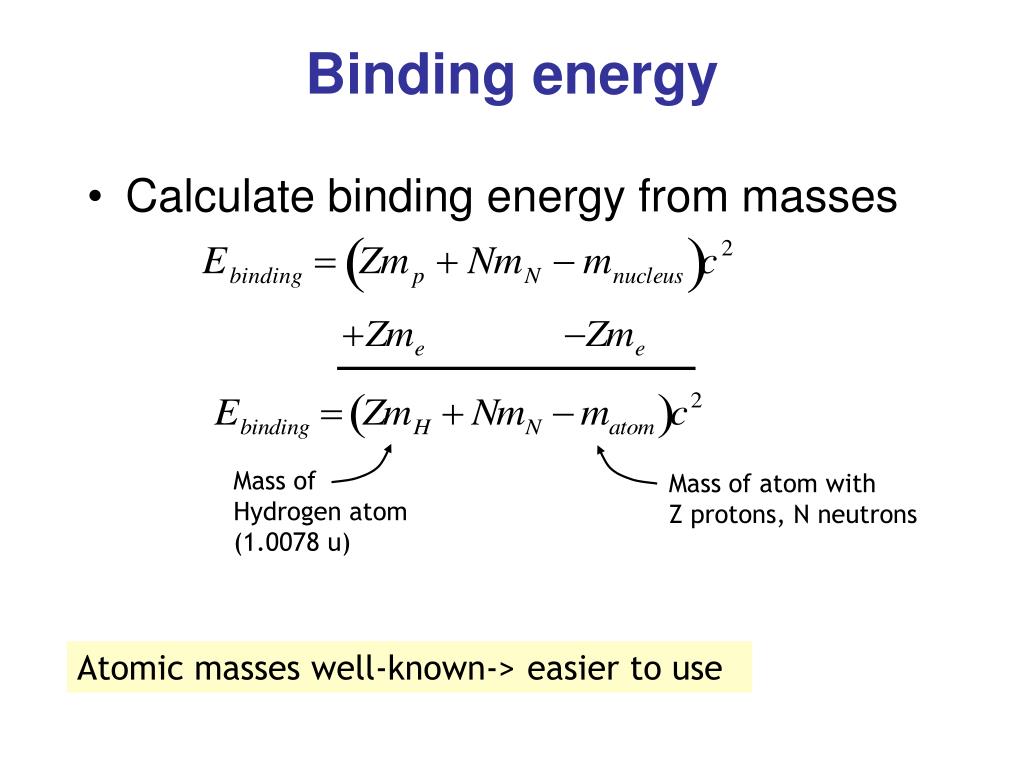
Taking into account the motion of the nucleus of a hydrogen atom, the expressions the electron's binding energy in the ground state and the Rydberg constant. How much (in per cent) do

5. In a hydrogen atom, the binding energy of the electron in the nth state is E. then the frequency of revolution or the electron in the nth orbits is: (A) 2E, /

Color online) Binding energy per atom of the onedimensional hydrogen... | Download Scientific Diagram

A hydrogen atom in a state having a binding energy of 0.85 eV makes transition to a state with - YouTube

If the binding energy of 2nd excited state of hydrogen like sample is 24ev approx,then determine the atomic number of - Chemistry - Structure of Atom - 12877143 | Meritnation.com

In a hydrogen atom, the binding energy of the electron in the ground state is `E_(1)` then - YouTube
If the binding energy of the electron in the ground state of hydrogen atom is E, then the frequency of electron in the nth orbit is

A hydrogen atom in a state having a binding energy (this is the energy required to remove an electron) of 0.85 eV makes a transition to a state with an excitation energy (

According to Bohr's theory of hydrogen atom , the product of the binding energy of the electron ... - YouTube

The binding energy of e in ground state of hydrogen atom is 13.6 eV. The energies required to out an electron from three lowest states of He ion will be - (in

The binding energy for the nth hydrogen atom by the most stable vac +... | Download Scientific Diagram