Groups identified by multi-IgG-ACE2-RBD correspond to defined ranges of... | Download Scientific Diagram
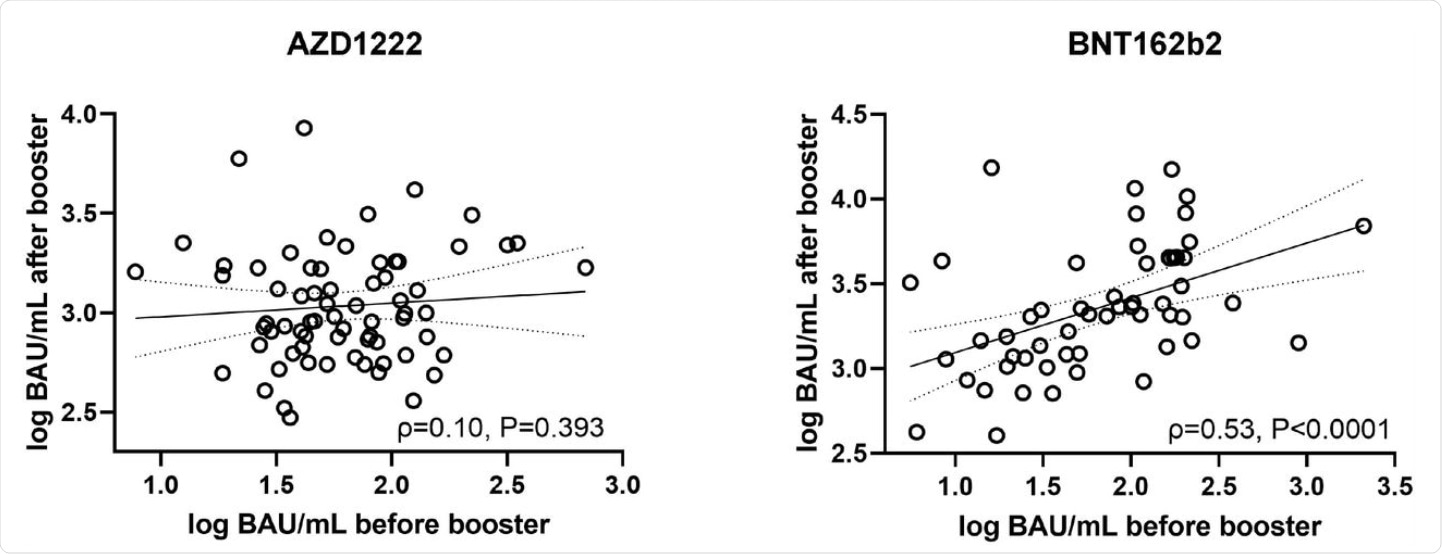
![PDF] Long-Term Humoral Immune Response against SARS-CoV-2 after Natural Infection and Subsequent Vaccination According to WHO International Binding Antibody Units (BAU/mL) | Semantic Scholar PDF] Long-Term Humoral Immune Response against SARS-CoV-2 after Natural Infection and Subsequent Vaccination According to WHO International Binding Antibody Units (BAU/mL) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2d61c22b4be5197f326fe02e23fee1c1129f7e29/5-Figure1-1.png)
PDF] Long-Term Humoral Immune Response against SARS-CoV-2 after Natural Infection and Subsequent Vaccination According to WHO International Binding Antibody Units (BAU/mL) | Semantic Scholar
Comparison of the binding antibody units per milliliter (BAU/mL) ratio... | Download Scientific Diagram

Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection | Nature Medicine

Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection | Nature Medicine
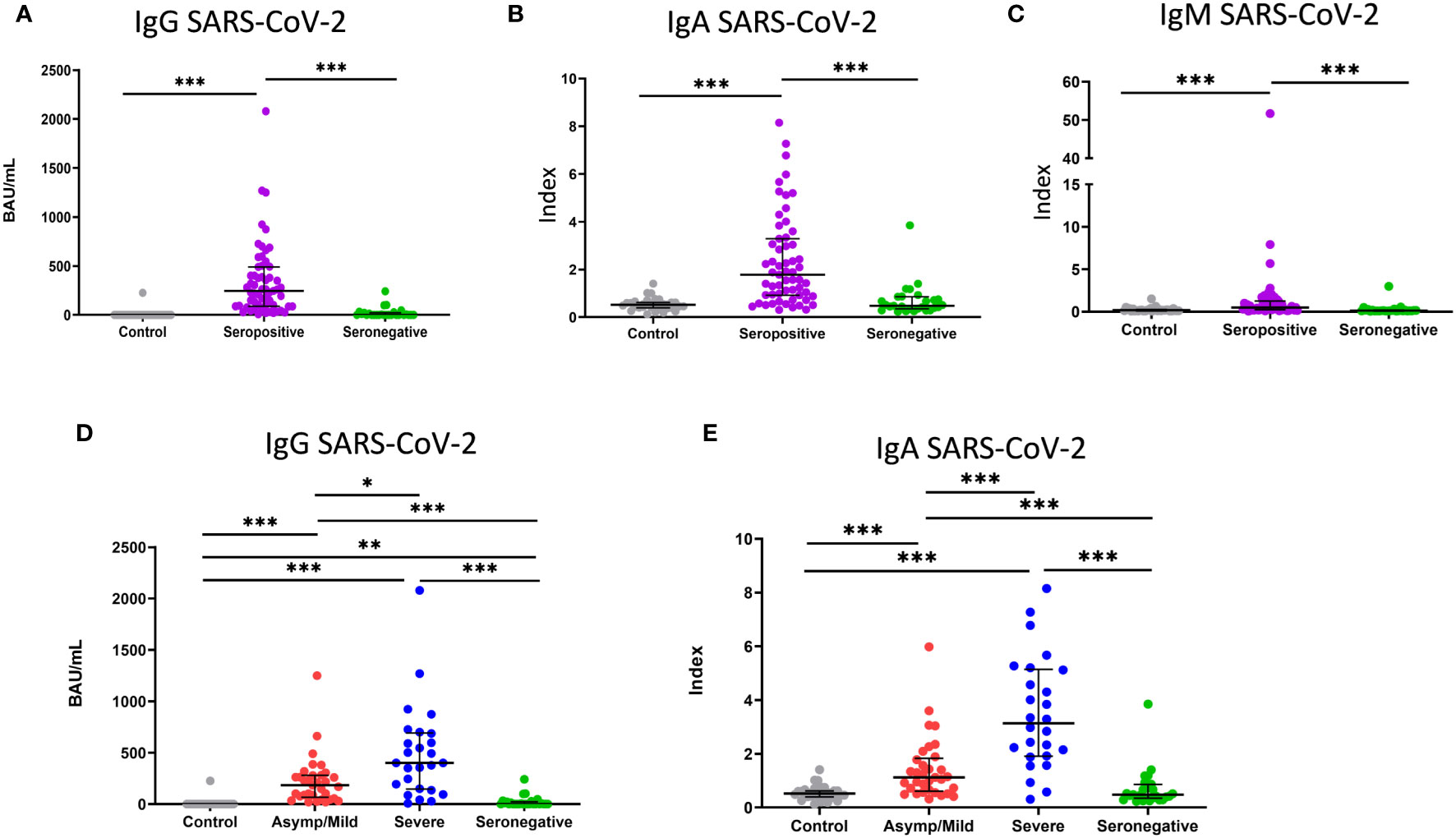
Frontiers | Memory SARS-CoV-2 T-cell response in convalescent COVID-19 patients with undetectable specific IgG antibodies: a comparative study
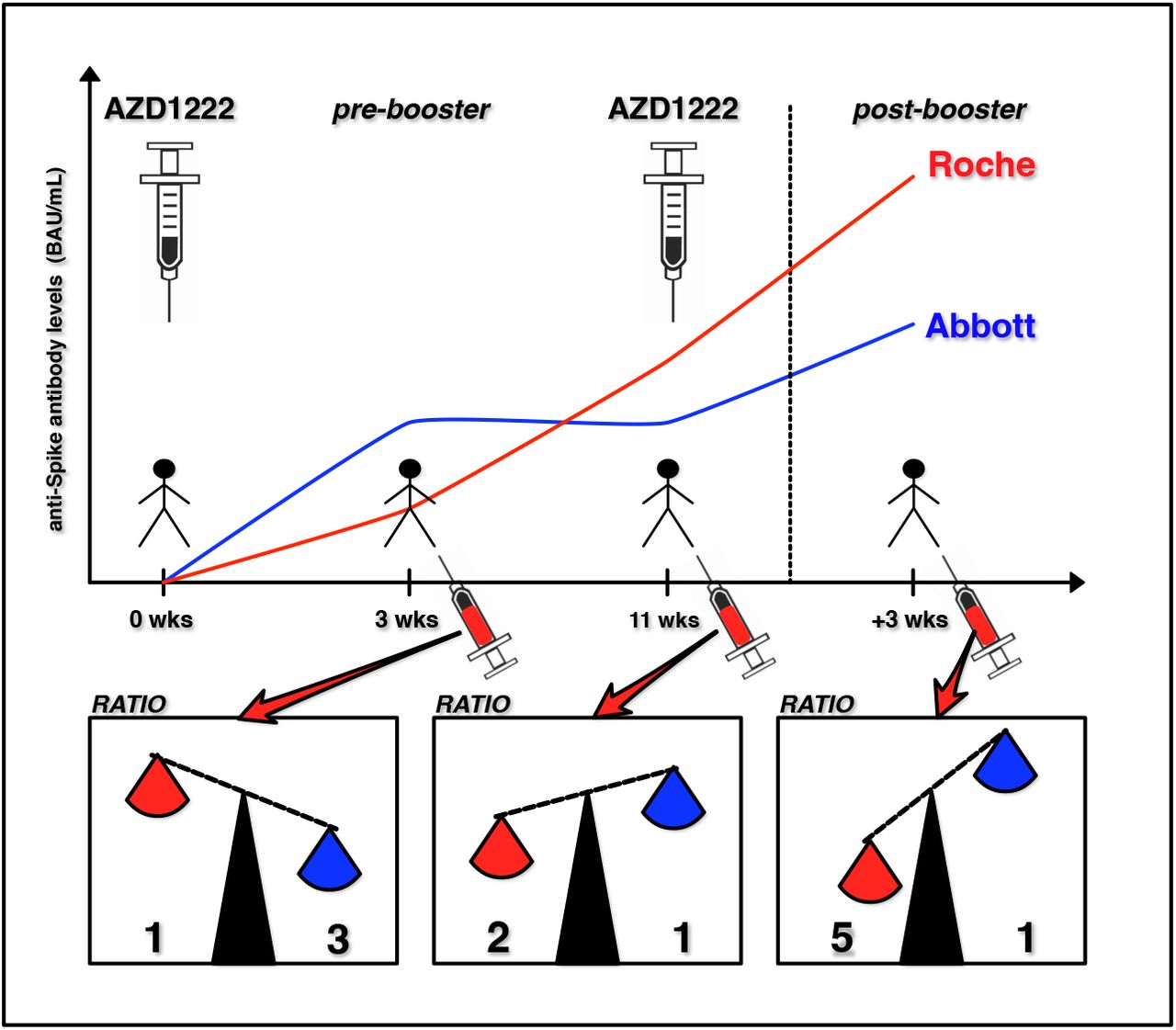
The comparability of Anti-Spike SARS-CoV-2 antibody tests is time-dependent: a prospective observational study | medRxiv

Multiplexed measurement of binding- and neutralizing antibodies to SARS-CoV-2 variants in 12.000 post-vaccine sera | bioRxiv

SARS-CoV-2 vaccine response and rate of breakthrough infection in patients with hematological disorders | Journal of Hematology & Oncology | Full Text
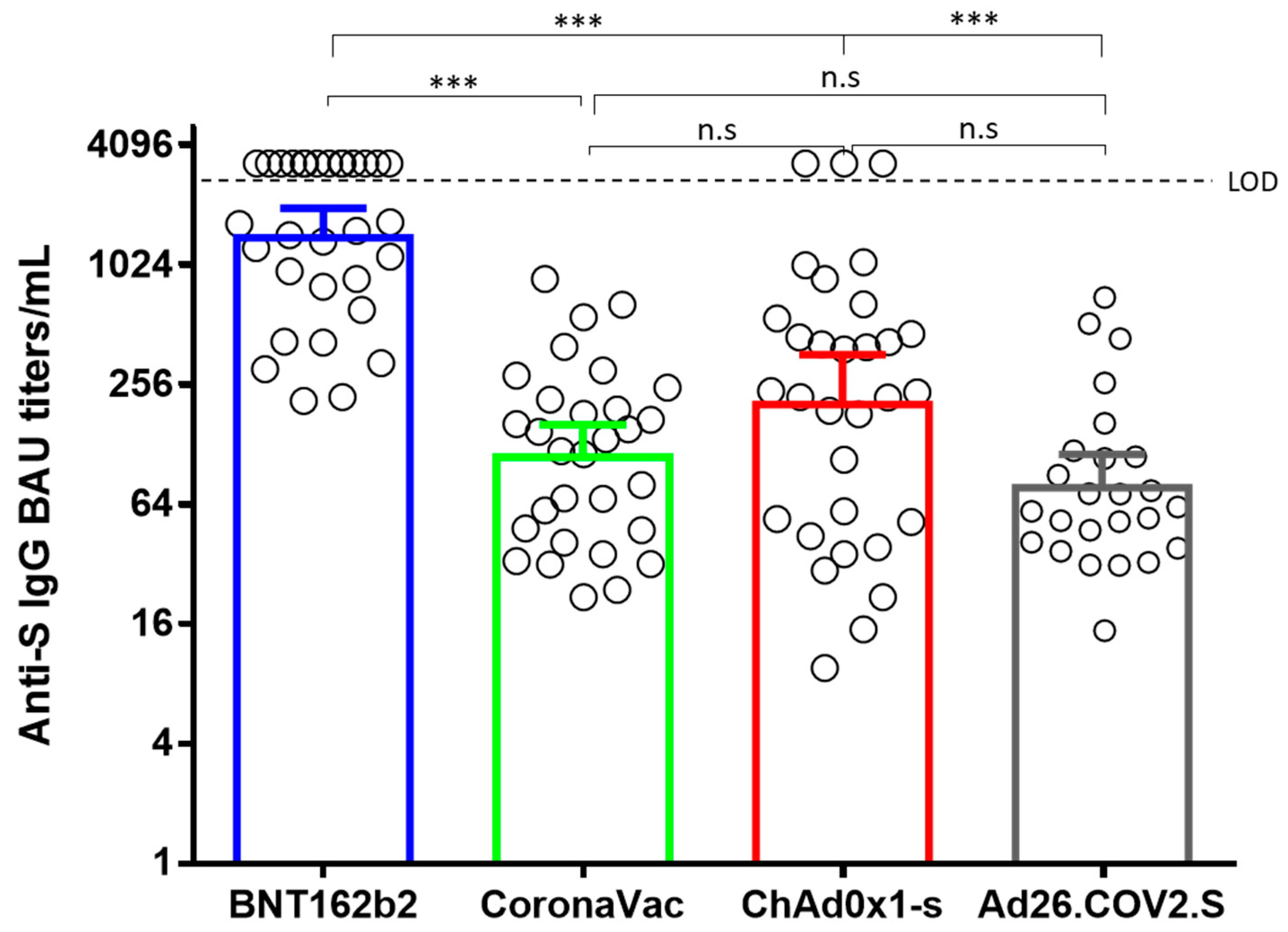
Vaccines | Free Full-Text | Neutralizing Antibody Responses Elicited by Inactivated Whole Virus and Genetic Vaccines against Dominant SARS-CoV-2 Variants during the Four Epidemic Peaks of COVID-19 in Colombia

Evolution of antibody responses up to 13 months after SARS-CoV-2 infection and risk of reinfection - eBioMedicine

Comparative Effectiveness and Antibody Responses to Moderna and Pfizer-BioNTech COVID-19 Vaccines among Hospitalized Veterans — Five Veterans Affairs Medical Centers, United States, February 1–September 30, 2021 | MMWR

Anti‐SARS‐CoV‐2 IgG antibody levels among Thai healthcare providers receiving homologous and heterologous COVID‐19 vaccination regimens - Kittikraisak - 2022 - Influenza and Other Respiratory Viruses - Wiley Online Library

Full article: Decay rate of antiS1/S2 IgG serum levels after 6 months of BNT162b2 vaccination in a cohort of COVID-19-naive and COVID-19-experienced subjects

SARS-CoV-2 Convalescent Sera Binding and Neutralizing Antibody Concentrations Compared with COVID-19 Vaccine Efficacy Estimates against Symptomatic Infection | Microbiology Spectrum

Quantitative measurement of IgG to SARS‐CoV‐2 antigens using monoclonal antibody‐based enzyme‐linked immunosorbent assays - Sander - 2022 - Clinical & Translational Immunology - Wiley Online Library
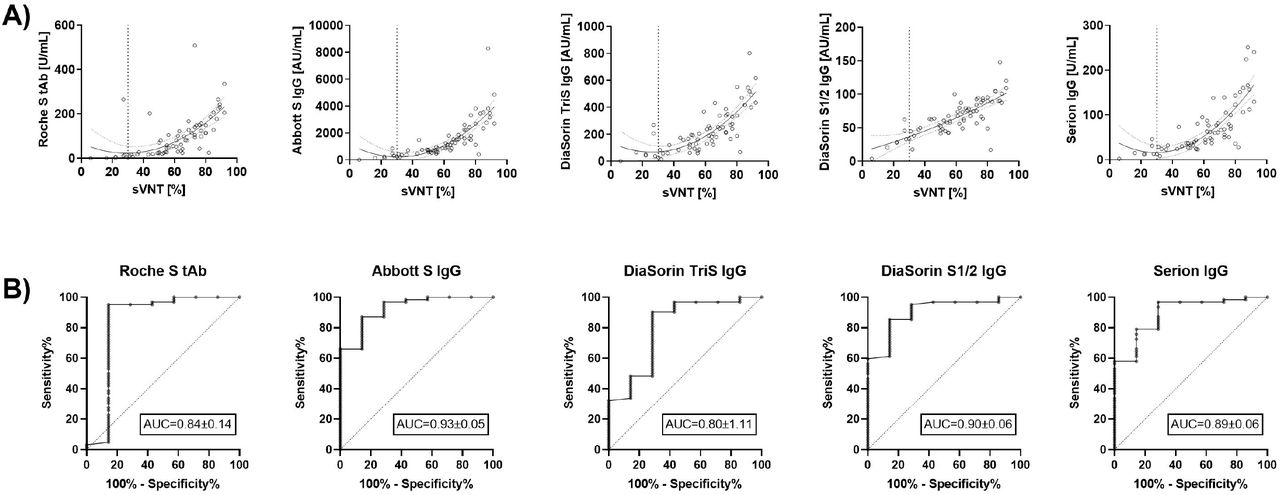
Anti-Spike protein assays to determine post-vaccination antibody levels: a head-to-head comparison of five quantitative assays | medRxiv

Kinetics of naturally induced binding and neutralizing anti-SARS-CoV-2 antibody levels and potencies among Kenyan patients with diverse grades of COVID-19 severity | medRxiv

Longitudinal data on humoral response and neutralizing antibodies against SARS-CoV-2 Omicron BA.1 and subvariants BA.4/5 and BQ.1.1 after COVID-19 vaccination in cancer patients | Journal of Cancer Research and Clinical Oncology

Anti-Spike Protein Assays to Determine SARS-CoV-2 Antibody Levels: a Head-to-Head Comparison of Five Quantitative Assays | Microbiology Spectrum